

Electric Fields
Coulomb's Law Remember, UNLIKE charges attract,

or
and LIKE charges repel,
or
What determines the magnitude of this electric force F?
The force F is directly proportional to the charges Q and q,
F Q q
The force F is inversely proportional to the square of the distance r between the charges,

F
1 / r2

F
1 / r2

F
1 / r2
We can combine these as
F Q q / r2
While proportionalities are good for qualitative discussions and comparisons, equations are much easier to use for calculations. We can change this to an equation with a constant of proportionality, k.
F = k Q q / r2 This constant of proportionality, k, depends upon the units we use. If we measure force F in newtons (N), distance r in meters (m), and charges Q and q in coulombs (C), then k has a value of
k = 9 x 109 N m2/C2 Now we must ask, just what is a coulomb of charge, anyway?
The electric charge of an electron or a proton is labeled e and is equal to . . .
Coulomb's Law describes the force F between two electric charges, Q and q, a distance r apart,
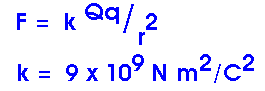
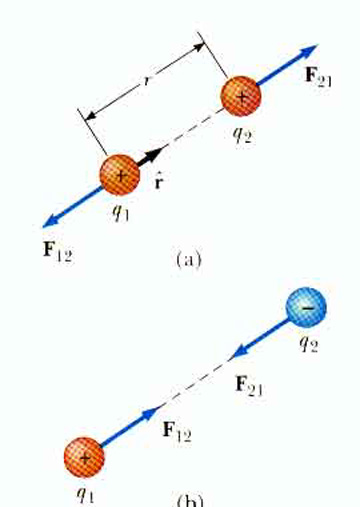
What is the force between two positive charges, 1
C and 2
C, when they are separated by a distance of 1 m?
Remember, force is a vector. Coulomb's Law lets us calculate the magnitude of the electric force but we must still keep in mind that force is a vector!
Example of Coulomb's Law We will also sometime write this "Coulomb's constant" as
where "epsilon-zero" is
and is known as the "permitivity of free space". That name sounds more ominous than it needs to. It is simply another form of "Coulomb's constant" -- and nothing more! There are major situations or major equations where we end up with a factor of
which is then more convenient to write as
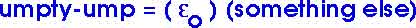
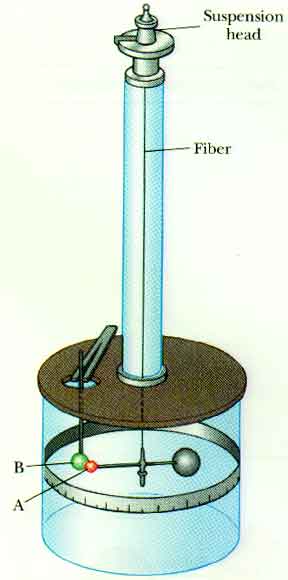
You may have looked at the Cavendish Balance when you talked about Gravity. Cavendish used a very delicate balance to measure incredibly small forces. Coulomb, in measuring the electrical forces, had larger forces to work with. Here is a sketch of the balance he used to measure electrical forces and determine the one over r-squared characteristic of the electrical force:
(c) Doug Davis, 2002; all rights reserved
Return to Ch23 ToC